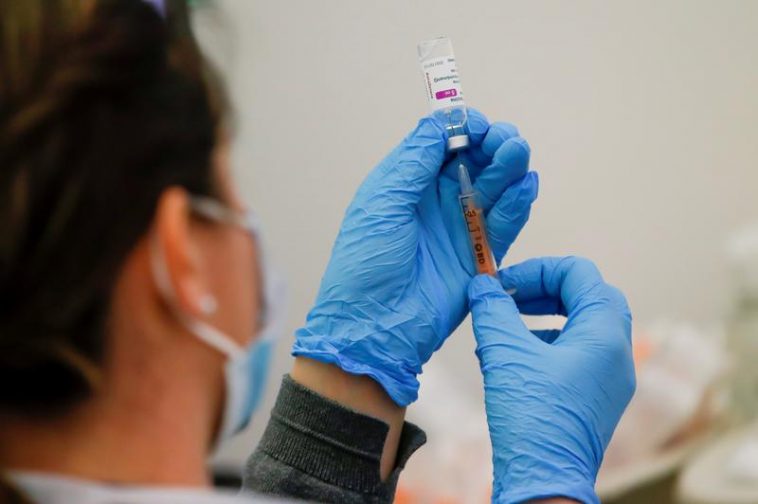
UK regulators mentioned on Thursday that they had identified 30 rare cases blood clots after receiving AstraZeneca COVID-19, 25 more than previously reported by the agency.
The Medicines and Healthcare merchandise Regulatory Agency (MHRA) mentioned that he had no such clotting experiences following injection of a vaccine manufactured by BioNTech SE and Pfizer Inc.
On Friday, a drug regulator told the Financial Times and The Guardian that seven AstraZeneca recipients had died after recording rare blood clotting events.
The MHRA, the European Medicines Agency, and the World Effectively being Group will confidently reiterate that the benefits of the vaccine in preventing COVID-19 outweigh any possible problems associated with blood clots.
Some international locations ban AstraZeneca, while others lead to resumption of vaccination.
On March 18, the UK medicines regulator mentioned that there were five cases of rare blood clotting out of the 11 million swabs administered.
Thursday is credited with 22 experiences of venous sinus thrombosis, an extremely rare condition associated with brain clotting, and eight experiences of alternative clotting events associated with low platelet counts out of the full 18.1 million doses administered.
for the easiest on the phone on-tablet-portrait-up for a tablet landscape for-desktop-up for-extensive-desktop-up



Comments
Loading…